 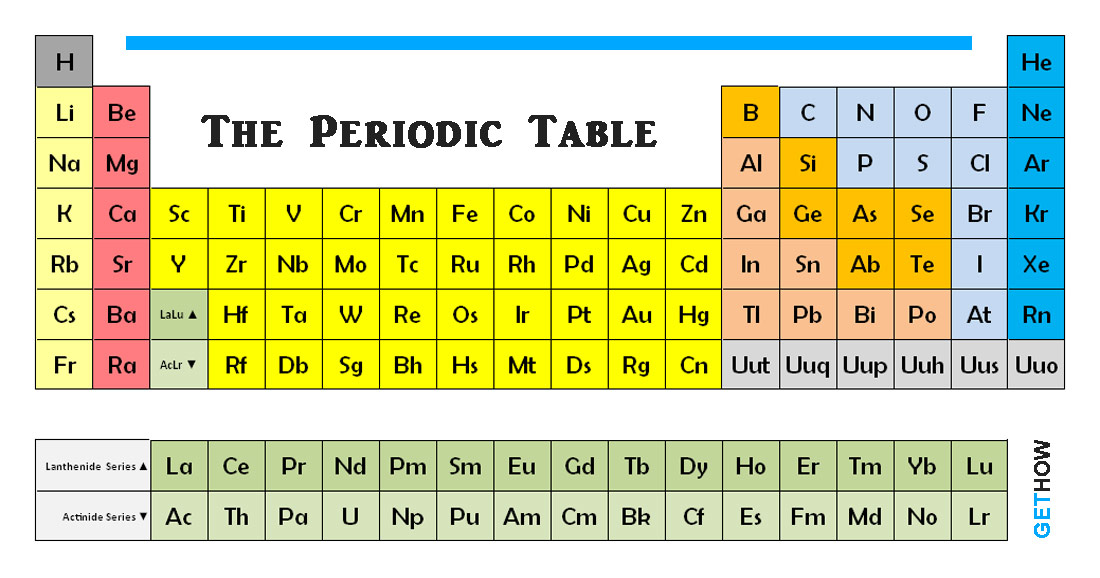 
The metals in group 1, but you don't find these Metals are reactive- not quite as reactive as Things like magnesiumĪnd calcium and strontium are your alkaline earth metals. So let me go ahead andĭraw that in green here. Let's talk about hydrogen,īecause hydrogen is also in group 1, but hydrogen Nature in combination with other elements. Walk outside and find some sodium lying on the ground. Going to find them in their pure state in nature. Turn out to be so reactive that you're not Organizing elements into groups is elements in the same Soft, silvery metals that are extremely reactive. In group 1, or group 1A, so things like lithium,Īnd sodium, potassium. Room, and we're not really going to talk about all With numbering my periods, so this would be So lithium, beryllium, boron, carbon, nitrogen, oxygen,įluorine, and neon. Hydrogen is in the first period and so is helium. Period 1, and I just move across my periodic table, Numbering your groups is useful when you're thinkingĪbout valence electrons. This group 3A, group 4A, group five 5A, groupĦA, 7A, and finally 8A. Groups 3 through 12, continue on with your That group 1 is group 1A, group 2 is group 2A. To number your groups, and that would be to say Up to here, and I can see I have another verticalĬolumn, so group 13, 14, 15, 16, 17, and finally 18. So all these elementsĪre in the same group, and we call this group 2. Of these elements are also in the same column. So all these elementsĪre in the same group. See that all of these elements are in the same vertical column. The groups are the verticalĬolumns on the periodic table. This types of changes involve the nuclei of atoms and are therefore in the realm of nuclear chemistry which is a more specialized field of chemistry which is rarer to encounter. Anytime you want to change an atom into a new element you must change the number of protons in the nucleus either by adding or removing them. Since our new nucleus has a different number of protons than the original nuclei, we have created a new element different from the original two. This is the most common case you will encounter in chemistry.ģ) At very high energies we can get the atoms of different elements moving so fast so that when they collide their nuclei can fuse into one larger nucleus. With a chemical reaction even though chemical bonds have been broken and reformed to create a new chemical, we haven't touched the protons in the nucleus and therefore haven't changed the elements involved. Which as we know is a chemical compound composed of the elements hydrogen and oxygen. Using the hydrogen and oxygen gas example, if we added those gases together and introduced a source of heat like a flame, they would react and combine to form water. Now there is enough energy to break chemical bonds and conduct a chemical reaction where the atoms of different elements can combine into a compound composed to atoms from both elements. For example, if I combine hydrogen and oxygen gas together at low temperatures then I just have a vessel filled with unreacted gas.Ģ) At higher energies we start creating the situation suitable for reactions to occur. You just get a mixture of different types of atoms. Combining two atoms of different elements together depends on how much energy you put into such a collision.ġ) At low energies (think low temperatures essentially) putting together two samples composed of atoms of different elements doesn't usually create a reaction. So in practice we're not really combining elements rather atoms of different elements. So elements are different types of atoms which are differentiated by their atomic number (the number of protons).
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
